
We think about studies differently.
We don’t count the number of edit checks. Nor do we bill for each query issued.
Axiom’s DM approach is designed to ensure that the resulting data is accurate, scientifically credible, and was thoroughly reviewed against the protocol, GCP and therapeutic requirements.
This is achieved through a multi-threaded approach prior to study launch and during the study operational phase.

The Data Management Timeline Starts Early
and Moves Forward with Constant Thinking
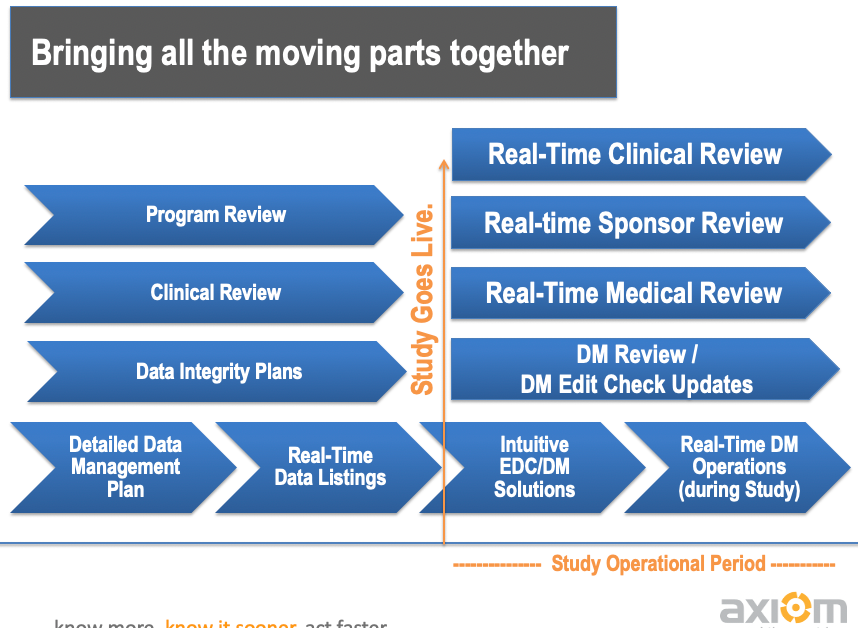
Data Review Tools Take All Forms.
At Axiom, We Focus on Ensuring that Any Aspect of the Data Can Be Easily Accessed and Reviewed.


These tasks represent the key areas that Axiom focuses on when developing a DM Plan:
Client Discussions on Protocol, Data Management Plan Structure
Regular discussions to ensure the protocol/therapeutic requirements are understood and the approach to the DM plans reflects the study needs.
Research / Review of Therapeutic Area
Team review of therapeutic area by lead data manager and supporting team members
Interactive Development of DM Plan
Plan development with regular iterations to client for review/discussion
Incorporation of lessons learned from previous studies / DM Plans
Review of Con Meds / Other Agents
Review of con meds / other agents that are permitted and/or prohibited by study subjects
Test Case Scenarios
Development of test case scenarios
Possible review of retrospective subjects to test against DM plan
DM Operations – Live Study
These tasks represent the key elements that Axiom focuses on to ensure high quality, scientifically credible study data:
Live Data Review against DM Plan
Ongoing data review based upon data entry and key milestones
Automated Edit Checks for System Data review
System review of key data points with results reported to Data Manager
Site Communication for Queries, Missing Information & other DM items
Ongoing site communication for DM related tasks
Regular client reporting for DM activities
Live Data Management Plan: Ongoing Review / Changes
Regular DM Plan reviews and possible changes
Update Discussions with Client on DM Plan Changes
Updates with clients on possible DM Plan changes, notations, and critical items to flag
Better decisions. Shorter timelines.
Unified Technology. Unmatched Service.

